 This ultimately resulted in a delayed pulsatile release profile. Once the shell had sufficiently degraded, it lost its barrier function, which caused the antigen to diffuse out of the implant. Initially, the polymeric shell formed an impermeable barrier to the in vitro release of the antigen, thereby resulting in a lag phase during which no antigen was released. This implant contained ovalbumin as a model antigen in a core that was surrounded by a non-porous shell of the biocompatible and biodegradable polymer poly( dl-lactide- co-glycolide) (PLGA). In a previous proof of concept study, the feasibility of a single-injection vaccine using a polymeric core-shell implant (oblong: ≈ 9 × 5 × 5 mm) was investigated. Therefore, the development of a formulation providing a delayed pulsatile release is the focus of this study. However, the development of the booster part of such a formulation, characterized by a pulsatile release after a predefined lag time, is challenging. The administration of the primer dose can easily be achieved by co-injection of a solution of the antigen or by the addition of a separate immediate-release formulation of the antigen. Such a pulsatile release formulation could provide for a prolonged immunological response, hence circumventing the need for multiple injections. The latter could be prevented by developing a single-injection vaccine formulation that exhibits a pulsatile release profile and thus includes both the primer and the booster doses. An example of a prime-boost vaccine is the diphtheria-tetanus-pertussis (DTP) vaccine of which, in 2020, 17.1 million infants did not receive a primer dose, and an additional 5.6 million were only partially vaccinated. Such a prime-boost schedule does not only cause logistical problems and high costs, it is also very uncomfortable and thus jeopardizes the compliance of the vaccinee. A multiple-injection schedule generally consists of a first immunization (primer) followed by a second or even third immunization (booster) after a certain period of time. One of the reasons for this low coverage is the limited access to routine immunization services, which is mainly a problem when multiple injections are required to obtain protective immunity against the targeted pathogens. 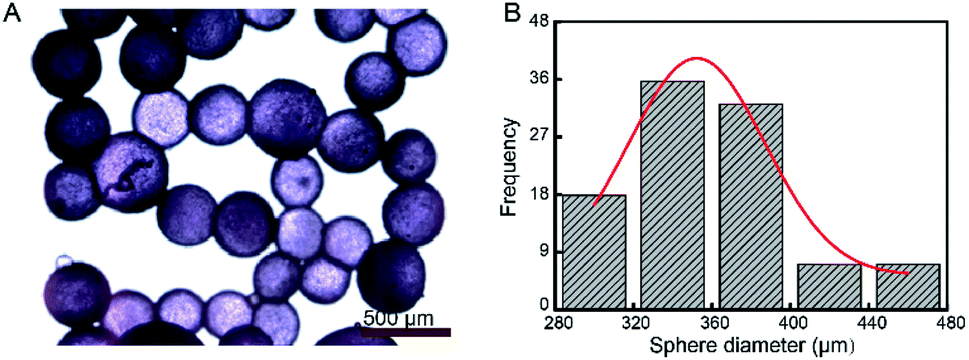
However, the global coverage for many vaccines is still too low, especially in low-income countries. Immunization is widely recognized as one of the greatest and most successful medical advances of the past centuries, saving two to three million lives every year by preventing or even eliminating infectious diseases. Altogether, these findings show that injectable core-shell microspheres combined with a primer are a promising alternative for the current multiple-injection vaccines. Co-injection of the core-shell microspheres together with a solution of the antigen that serves as the primer would allow for the desired biphasic release profile. Shell thickness and bovine serum albumin loading had no effect on the release behavior, which could be ascribed to the degradation mechanism of the polymer, with bulk degradation being the main pathway. This lag time increased with the increasing lactide content of the polymer and ranged from 3 to 7 weeks. All formulations exhibited a pulsatile in vitro release of BSA after an adjustable lag time. Highly monodisperse particles with different particle characteristics were obtained using a microfluidic setup. These microspheres contained bovine serum albumin (BSA) as the model antigen and poly( dl-lactide- co-glycolide) (PLGA) with various dl-lactide:glycolide monomer ratios as the shell material. In this study, we developed parenterally injectable core-shell microspheres with a delayed pulsatile release profile that could serve as the booster in such a vaccine formulation. 
A single-injection vaccine formulation that provides for both administrations could greatly improve the convenience and vaccinee’s compliance. These injections often consist of a primer administration followed by a booster administration of the vaccine a few weeks or months later. For many vaccines, multiple injections are required to confer protective immunity against targeted pathogens. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
